Cervical Screening
This section contains the clinical practice guidelines and clinical pathways for cervical cancer screening in Alberta.
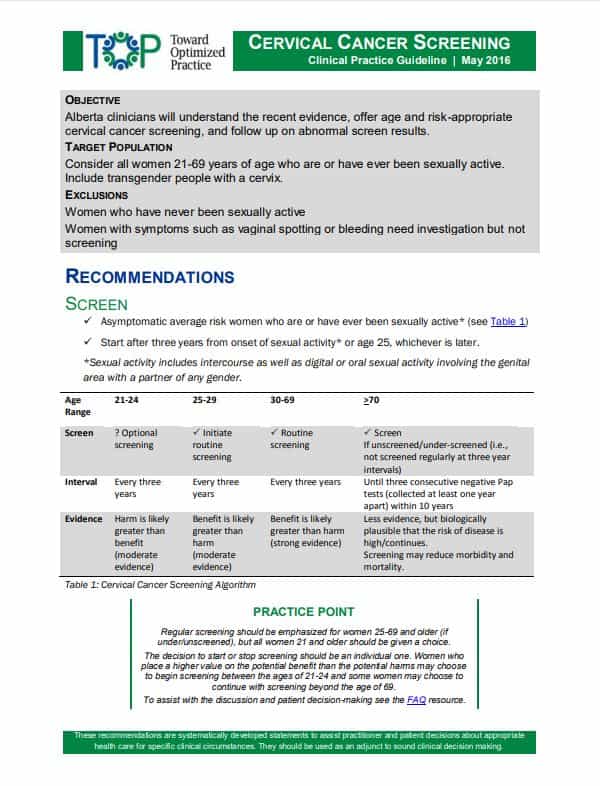
2016 TOP Clinical Practice Guidelines
View the PDF of the clinical practice guidelines.
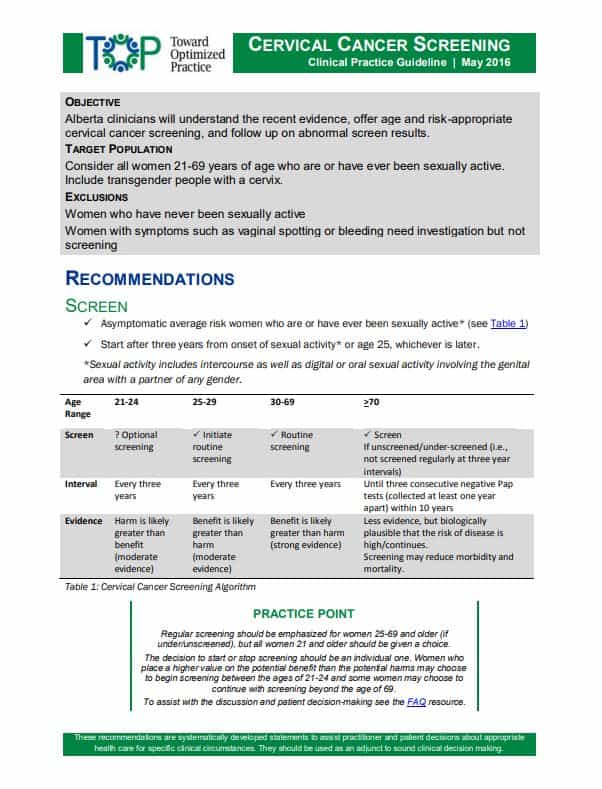
2016 TOP Clinical Practice Summary
View the PDF of the clinical practice guidelines summary.
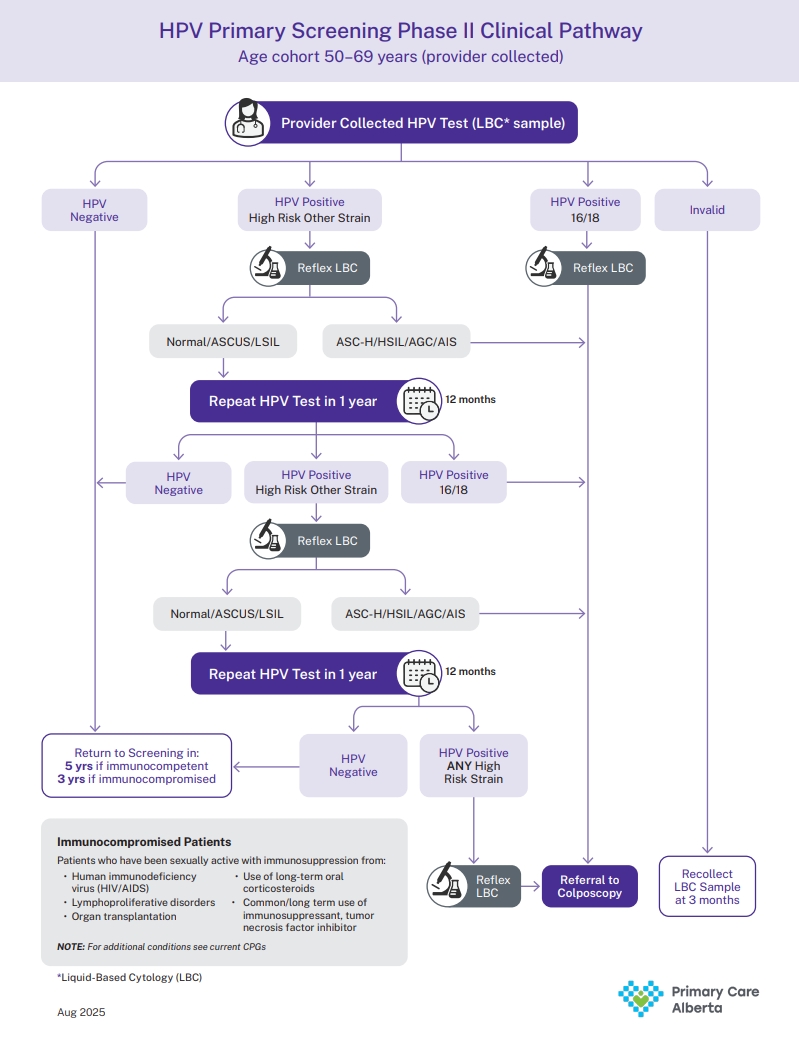
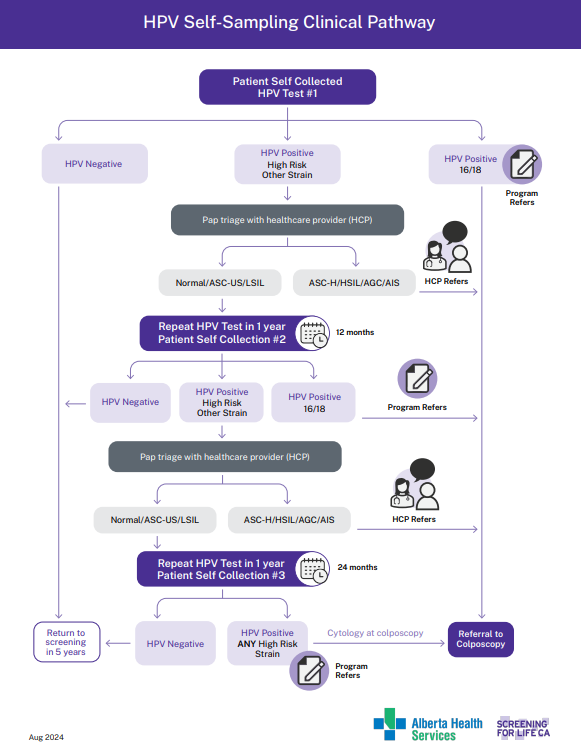
HPV Primary Clinical Pathway
View the PDF of the HPV Clinical Pathway Algorithm.
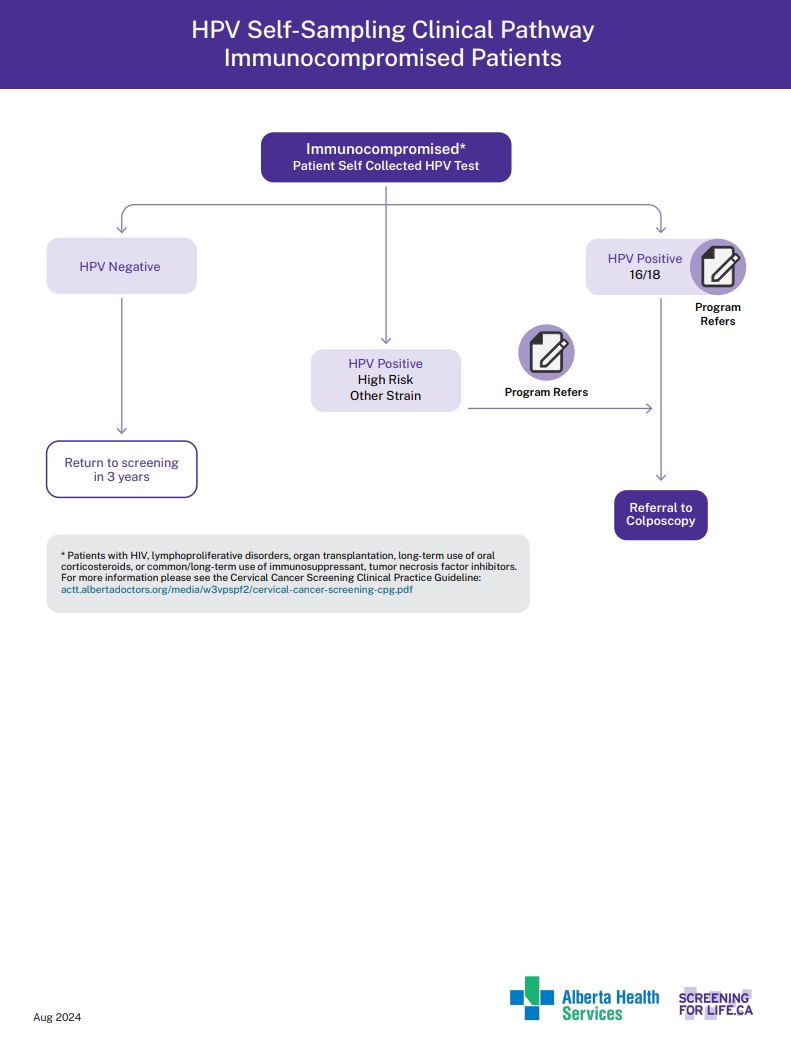
HPV Self-Sampling Clinical Pathway Immunocompetent Patients
View the PDF of the HPV Self-Sampling Clinical Pathway Immunocompetent Patients.
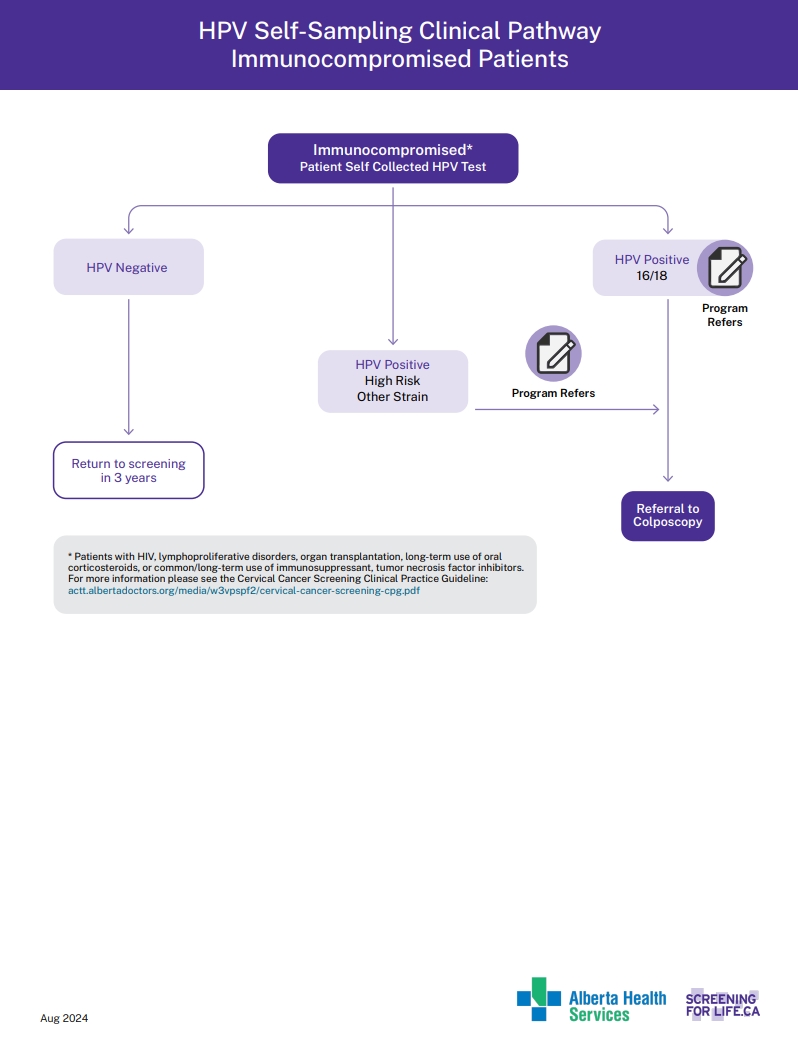
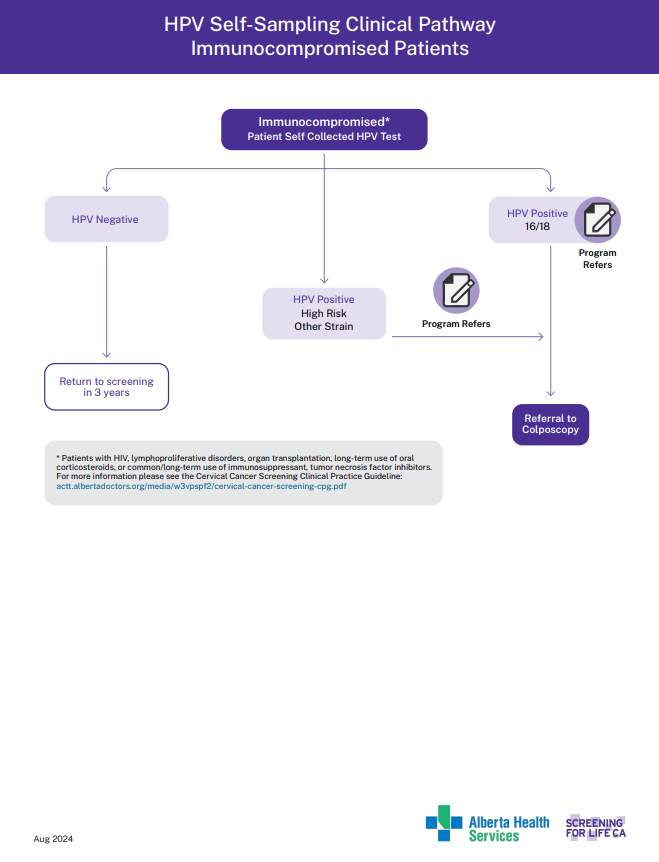
HPV Self-Sampling Clinical pathway Immunocompromised Patients
View the PDF of the HPV self Sampling Immunocompromised.
This section contains information for Registered Nurses (RNs) performing Cervical Cancer Screening.

ACCSP Cervical Cancer Screening Provided by Registered Nurses Guideline
*UPDATED July 7, 2020. This document guides RNs through the cervical cancer screening process

CRNA Practice Advice: Registered Nurse role in cervical cancer screening
This document outlines the role of Registered Nurses in Cervical Cancer Screening

ACCSP Cervical Cancer Screening Learning Module for Registered Nurses (2022)
*UPDATED June 15, 2022.
This section contains information and resources for when your patient has an abnormal Pap result, and resources for colposcopy.
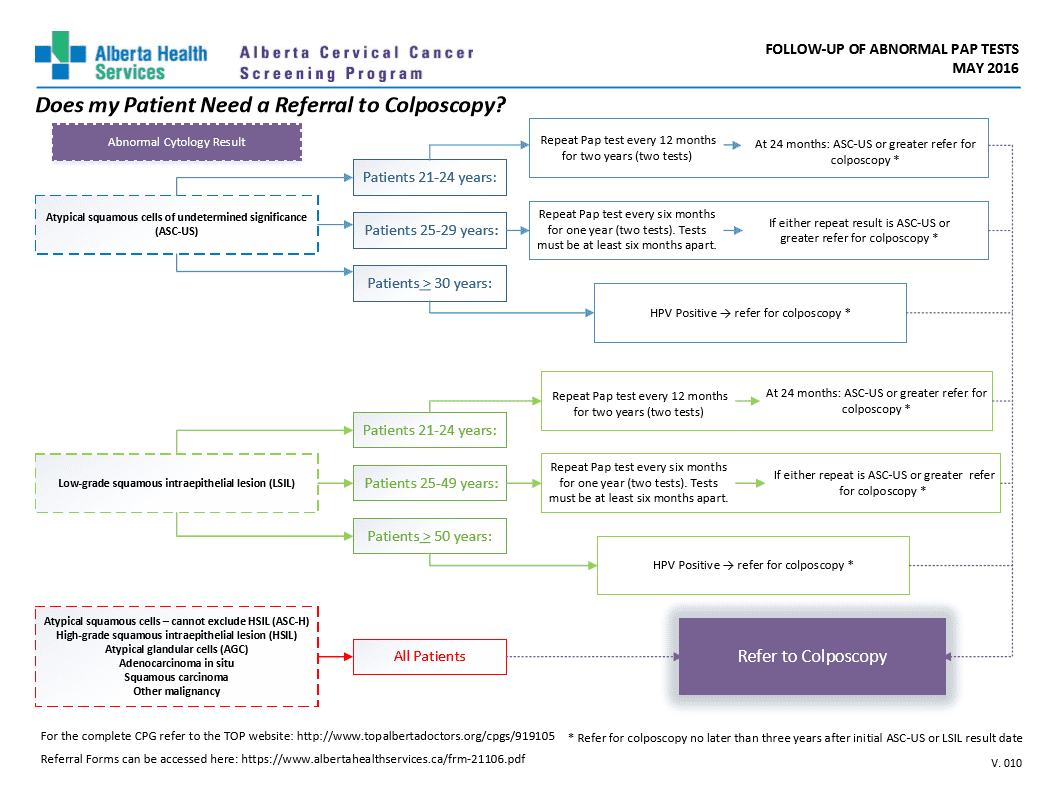
Does your patient need a referral to colposcopy?
This algorithm will help you determine if your patient needs a referral to colposcopy.

Information for healthcare providers about reflex HPV testing
Find out more about HPV reflex testing and what it means for your patient.

Request form for completed colposcopy record
Use this form if you are requesting a patients colposcopy record.
Brochure
A Woman’s Guide to Laser Surgery
Booklet
ACCSP Cervical Screening Provided by RNs Guidelines
Info Sheet
ACCSP HPV ToC Information for Health Care providers
Brochure
Arabic - Cervix Self-Screening
Brochure
Arabic - Cervix Self-Screening Instructions
Brochure
Blackfoot Cervix Self-Screening
Brochure
Blackfoot Instructions Community
Other
CC1762 CMIO Colpo Manual March 2024
Brochure
Cervical Cancer Screening for the 2SLGBTQI+ Community
Brochure
Cervix Self-Screening Instructions
Brochure
Cervix Self-Screening Pilot Project Brochure
Brochure
Cervix Self-Screening Results
Brochure
Chinese Cervix Self-Screening
Brochure
Chinese-Cervix-Self-Screening Instructions
Brochure
Clinical Practice Guidelines - Cervical
Brochure
Clinical Practice Guidelines Summary - Cervical
Brochure
Colpo 2022 Annual Report
Info Sheet
Colposcopy Frequently Asked Questions
Brochure
Colposcopy Information Guide
Booklet
Colposcopy Quality Improvement Guidelines
Brochure
Colposcopy webpage survey October 2023
Booklet
CRNA Practice Advice: Registered Nurse role in cervical cancer screening
Brochure
Dari Cervix Self-Screening
Brochure
Dari Cervix Self-Screening Instructions
Brochure
Dec 13, 2023 ACCSP Colpo QI Minutes
Brochure
Dellino, M. et al, 2022, Lactobacillus Crispatus M247 Oral Administration Effective in Management of Papillomavirus-infected Women
Brochure
Denesuline Cervix Self-Screening
Brochure
Denesuline Instructions Community
Brochure
Desravines, N. et al, 2020, Topical Therapies for the Treatment of CIN 2-3
Brochure
Dhillon, N. et al, 2020, Bridging Barriers to Cervical Cancer Screening in Transgender Men
Brochure
Do I really need a Pap test?
Info Sheet
Does my patient need a referral to colposcopy?
Brochure
French Cervix Self Screening
Brochure
French Instructions Community
Brochure
Gilliland, K. et al, 2013, Cytology of Vulvar Vaginal Paget Disease
Brochure
Grootenhuis, N. et al, 2019, Margin Status Revisited in Vulvar Squamous Cell Carcinoma
Form
Healthcare provider form to report ineligibility for cervical screening
Brochure
HPV Reflex
Brochure
HPV Self-Sampling Clinical Pathway
Brochure
HPV Self-Sampling Clinical Pathway Immunocompromised Patients
Brochure
HPV Test of Cure
Info Sheet
HPV Test of Cure Algorithm- One Pager
Brochure
HPV Test of Cure Patient Resource
Info Sheet
HPV Test of Cure Patient Resource and HPV Vaccine
Info Sheet
HPV testing information for women having Pap tests
Booklet
HPV ToC Colposcopy Implementation Handout
Brochure
HPV-Primary-PCP_Overview-and-FAQ
Brochure
HPV-SS-Clinical-Pathway
Brochure
HPV-SS-Clinical-Pathway_Immunocompromised
Brochure
HPV: What You need to know and do
Brochure
Huang, S. et al, 2023, Effect of heat-clearing and Dampness-eliminating Chinese Medicine for High-risk Cervical Cancer Papillomavirus Infection
Info Sheet
Information for healthcare providers about reflex HPV testing
Other
Invite letter to the Alberta Cervical Cancer Screening Program
Brochure
June 1, 2023 ACCSP Colpo QI Minutes
Brochure
Kiran, T. et al, 2019, Cancer Screening Rates Among Transgender Adults
Brochure
Litwin, C. et al, 2021, High-risk HPV Prevalence Among Women Undergoing Cervical Cancer Screening (1)
Brochure
LOOP Electrosurgical Excision Procedure (LEEP) – Operating Room
Brochure
LOOP Electrosurgical Excision Procedure (LEEP) – Outpatient
Brochure
Luo, M. et al, 2019, Detoxification Therapy of Traditional Chinese Medicine for Genital Tract High-risk HPV Infection
Brochure
Making sense of abnormal Pap test results
Brochure
May 26, 2025 - Colpo Meeting minutes
Brochure
Micheletti, L. et al, 2016, Vulvar Lichen Sclerosus and Neoplastic Transformation
Brochure
PCP two pager ACCSP
Brochure
Plains Cree Cervix Self Screening
Brochure
Plains Cree Instructions Community
Brochure
Potter, J. et al, 2015, Cervical Cancer Screening for Patients on the Female-to-Male Spectrum
Brochure
Preti, M. et al, 2019, Clinical Role of Laser for Vulvar and Vaginal Treatments in Gynecology and Female Urology
Brochure
Preti, M. et al, 2022, ESGO, ISSVD, ECSVD, ECSVD and EFC Consensus Statements on Pre-invasive Vulvar Lesions
Brochure
Primary Care Provider Follow-Up Table
Other
Registered Nurse Pap test learning module
Form
Request form for completed colposcopy record
Brochure
Select Provider Frequently Asked Questions
Brochure
Serrano, L. et al, 2021, Efficacy of a Coriolus Versicolor-Based Vaginal Gel in Women with HPV Dependent Cervical Lesions
Poster
Should I have a Pap test?
Brochure
Smith, J. et al, 2022, AHCC Supplementation to Support Immune Function to Clear Persistent HPV Infections
Brochure
SOGC Guidelines HPV+ and Specific Populations
Brochure
SOGC Guidelines Risked based approach to cervical Dysplasia
Brochure
SP-HPVClinicalPathwayAlgorithm
Brochure
Spanish Cervix Self-Screening
Brochure
Spanish Cervix Self-Screening Instructions
Brochure
Thompson, E. et al, 2022, Significance of p53 and Presence of dVIN at Resection Margin in Early Stage HPV-independent Vulvar Squamous Cell Carci
Brochure
Tigrinya Cervix Self-Sampling
Brochure
Tigrinya Cervix Self-Sampling Instructions
Brochure
Ukranian Cervix Self-Sampling
Brochure
Ukranian Cervix Self-Sampling Instructions
Brochure
Vieira-Baptista, P. et al, 2022, Risk of Development of Vulvar Cancer in Women with Lichen Sclerosus or Lichen Planus
This section contains clinical information and resources for Colposcopy.

Colposcopy clinic resource for standardized patient care in Alberta

HPV Test of Cure Information for Primary healthcare providers

HPV Test of Cure Patient Resource and HPV Vaccine

HPV Test of Cure Colposcopy Implementation Handbook
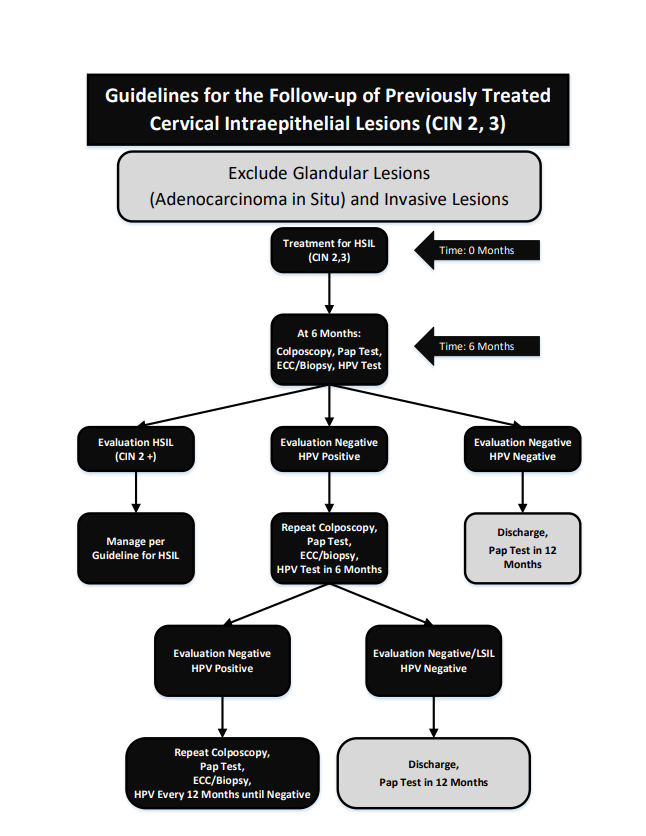
HPV Test of Cure Algorithm
This section contains information and resources for unsatisfactory results.
What does an unsatisfactory Pap result mean?
This means the test was rejected/not processed by the laboratory or that the specimen was processed and examined but was unsatisfactory for evaluation of epithelial abnormality or the cells could not be seen well enough to give a result.
Why is it important?
The ACCSP monitors quality and performance indicators in the cervical cancer screening pathway. One of these is the unsatisfactory rate. The provincial target is <1%.
Causes
Unsatisfactory Pap test results are mostly due to cervical sampling and specimen collection issues. This may be due to a number of factors which includes:
- Scant cellularity.
- Faulty technique.
- Post-menopausal status.
- Excess lubricant use.
- Interfering substances like mucus, blood and bacteria.
- Human error such as:
- Unlabeled specimens: Name and unique identifier missing.
- Incorrect or mislabeled specimens: For example, patient details (name or different patient ID) on requisition form and sample container don’t match.
- Identifier incomplete: Missing fields like first or last name missing, initials only, missed numbers on patient’s PHN.
- Significant misspelling where more than 2 letters are transposed or missing/added, letters that changes the interpretation of the name.
- The use of incorrect containers or preservatives.
- Test request received without a corresponding sample or vice versa.
- Sample container not properly sealed leading to loss of sample.
What happens after an unsatisfactory result?
Every unsatisfactory Pap leads to a repeat Pap test in 3 months.
What can I do to reduce my unsatisfactory rates?
Patient related:
- Book their appointments for a day they won’t be on their periods (but should book an appointment if they have an abnormal bleeding). Wait at least 7 – 10 days after first day of menstrual period.
- Prepare your patients for their Pap test by informing them about the following:
- Avoid douching or using contraceptive creams, vaginal medicines, sprays, powders or jellies in the 48 hours before the test.
- Avoid sexual intercourse in the 24 hours before the test.
- Avoid the use of personal lubricants in the 48 hours before the test.
- Avoid having a Pap test when you are being treated for cervical or vaginal infections. Wait at least 2 weeks after treatment ends before having a Pap test.
Physician related
- Collect samples far enough into endocervical canal to obtain cells.
- Follow manufacturer’s instructions for transferring the sample from the instrument to the liquid medium.
- Use lukewarm water for speculum lubrication.
- If clinically indicated or for patient’s comfort, use dime sized carbomer free lubricants.
- Provide all relevant clinical information on laboratory requisition forms.
Manufacturers’ guides
Resources
APL
- Sample Acceptance Policy: Acceptance of Laboratory Samples and Test Requests Policy (albertahealthservices.ca)
- Gyneacological Cytopathology Requisition Completion Aid: PCYSCR00003 Gynecological Cytopathology Requisition Completion Aid (albertahealthservices.ca)
- Label Placements: PA17-02.010 Laboratory Specimens – Label Placement (albertahealthservices.ca)
This section contains information on the HPV Testing for primary healthcare providers.
On this page:
- HPV Primary Screening
- HPV Self-Sampling (Cervix Self-Screening) Pilot Project
- HPV Test of Cure
- HPV Reflex Testing
—————————————————————————————————————————————————————————————————————
HPV Primary Screening
About HPV Primary Screening Implementation
Alberta is transitioning to the human papillomavirus (HPV) test as the province’s standard for cervical cancer screening. This is called HPV primary screening, also known as the cervical screening test. Until now, Pap tests have been used to screen for cervical cancer.
Phased approach
Starting on November 5, 2025, women and people with a cervix aged 50 to 69 years will be eligible for HPV primary screening. Beginning with this age group ensures access to a more sensitive test before these patients exit the cervical cancer screening pathway. Planning for provincial access to HPV test for women and people with a cervix aged 25 to 49 is ongoing and would occur at a later phase.
Why this change?
- HPV primary screening is more accurate, detects precancerous lesions earlier, reduces screening frequency, and allows for self-sampling, which reduces barriers to access for cervical cancer screening. This leads to improved patient access and health system efficiency. Planning for provincial access to self-sampling is also ongoing.
- This approach will address cytotechnologist workforce shortages, prolonged Pap turnaround times and colposcopy capacity due to anticipated surge in colposcopy referrals and volumes.
- By adopting HPV primary screening, Alberta will align with global and national efforts to eliminate cervical cancer by 2040.
Key Changes
| Aspect | Pap test | HPV primary screening |
| Eligibility (Age group) |
25 to 49 | 50 to 69 |
| Test at lab | Pap | High-risk HPV test |
| Screening interval | Every 3 years if normal Annual screens as per clinical practice guidelines |
Every 5 years (if HPV-negative and immunocompetent) Every 3 years (if HPV-negative and immunocompromised) |
| Sample collection | Provider-collected | Provider-collected (same liquid-based cytology (LBC) sample as per the current collection process) |
| Referral criteria | High-grade Pap: Refer to colposcopy Low-grade cytology and HPV reflex positive: Refer to colposcopy |
Positive for high-risk HPV types 16, 18: Refer to colposcopy Other high-risk strains: Refer based on HPV reflex cytology results |
What primary care providers need to do
Educate and reassure patients:
- To ensure consistency with program materials, primary care providers should use the term cervical screening test when referring to HPV primary screening. This is the name used in all patient resources.
- Explain the change in cervical cancer screening tests and the reasons behind it.
- Emphasize the improved sensitivity and safety of extended intervals.
- Address common concerns (see the FAQs below for more information).
Order the cervical screening test as before (No changes)
- The ordering process is the same as the current Pap collection in your clinic. At the lab, the HPV test is applied for patients aged 50 to 69. If a positive high-risk HPV strain is detected, a reflex cytology will be performed on the same sample.
- Use the same lab requisition (Gynecological Cytopathology Requisition) as per current process.
- On the requisition form, indicate if your patient is immunocompromised as that will impact the recommended screening intervals.
- See definitions for immunocompromised on the HPV primary clinical pathway.
Interpret HPV primary screening results
- Interpret results based on the HPV primary screening clinical pathway below.
HPV primary screening clinical pathway (updated Feb 26th, 2026)
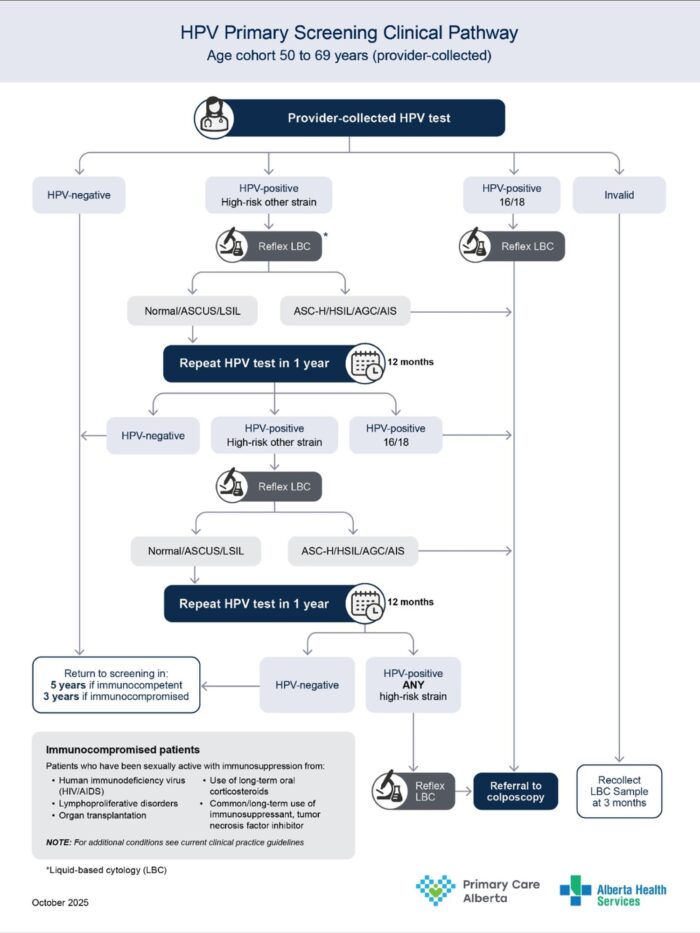
Download the HPV primary screening clinical pathway
Primary care provider results follow-up (updated Feb 26th, 2026)
| Result Group | HPV primary result (High-risk HPV NAT) |
Provider information |
| Normal | Not detected (aged 50 to 64) |
Repeat cervical cancer screening in 5 years. If immunocompromised, repeat in 3 years. |
| Not detected (aged 65 to 69) |
Routine screening no longer indicated for immunocompetent patients. Assess the patient as required for further screening. | |
| Abnormal | Detected HPV 16 and/or 18 + Unsatisfactory, NIL, ASCUS, LSIL, ASCH, HSIL, AGC, AIS |
Refer to colposcopy. Include the patient’s HPV and gynecological cytology (Pap test) results on the colposcopy referral form. |
| Detected other HR HPV (Immunocompromised) + Unsatisfactory, NIL, ASCUS, LSIL, ASCH, HSIL, AGC, AIS |
Refer to colposcopy. Include the patient’s HPV and gynecological cytology (Pap test) results on the colposcopy referral form. |
|
| Detected other HR HPV (Immunocompetent) + ASC-H, HSIL, AIS, AGC |
Refer to colposcopy. Include the patient’s HPV and gynecological cytology (Pap test) results on the colposcopy referral form. |
|
| Detected other HR HPV (Immunocompetent) + NIL, ASCUS, LSIL |
Repeat cervical cancer screening in 12 months (Rescreen to be completed via LBC sample). | |
| Detected other HR HPV (Immunocompetent) + Unsatisfactory Pap |
Repeat cervical cancer screening test in 3 months (Rescreen to be completed via LBC sample). | |
| Detected other HR HPV (Immunocompetent) + NIL, ASCUS, LSIL (after 2 previous results, each at least 12 months apart) |
Refer to colposcopy. Include the patient’s HPV and gynecological cytology (Pap test) results on the colposcopy referral form. |
|
| Other | First invalid result | Repeat cervical cancer screening test in 3 months (Rescreen to be completed via LBC sample). |
| Second consecutive invalid + ASC-H, HSIL, AIS, AGC |
Refer to colposcopy. Include the patient’s HPV and gynecological cytology (Pap test) results on the colposcopy referral form. |
|
| Second consecutive invalid + NIL, ASCUS, LSIL |
Repeat screening in 12 months (Rescreen to be completed via LBC sample). | |
| Indeterminate + ASC-H, HSIL, AIS, AGC |
Refer to colposcopy. Include the patient’s HPV and gynecological cytology (Pap test) results on the colposcopy referral form. |
|
| Indeterminate + NIL, ASCUS, LSIL |
Repeat cervical cancer screening in 12 months (Rescreen to be completed via LBC sample). | |
| Indeterminate + Immunocompromised + Any reflex cytology result |
Refer to colposcopy. Include the patient’s HPV and gynecological cytology (Pap test) results on the colposcopy referral form |
|
| Cancelled specimen | Repeat cervical cancer screening test in 3 months. |
Download the Primary care provider results follow-up table
Download pdf 
Implementation guidelines
Laboratory process:
- Samples will be collected from clinics as per current process.
- HPV test conducted as primary test for samples that meet the age eligibility (50 to 69 years).
- Reflex cytology is automatically done for HPV-positive, indeterminate and second consecutive invalid samples.
- Results sent as per current lab communication process with provider.
- Results can also be found in Netcare under Pathology (same location as Pap).
- HPV test and reflex gynecological cytology results will be reported separately. You may receive the results of the HPV test prior to the reflex cytology and on separate days.
Colposcopy referral Process
- There are no changes to the referral process.
- Clinics using FAST (Facilitated Access to Specialized Treatment) should continue submitting referrals through their respective Zone FAST Team. Click here for the Provincial Adult Gynecology Referral Pathway.
- Clinics that use the Alberta Referral Directory should continue to do so in accordance with their current practice.
Clinical considerations
Self-collection (also known as HPV self-sampling or cervix self-screening)
- The HPV Self-Sampling Pilot Project is no longer accepting new patients or orders for self-screening test kits.
- The purpose of the pilot was to increase cervical cancer screening access and participation in under-screened populations (Indigenous, newcomer and rural/remote).
- Future consideration: Self-sampling is part of the planning that is underway for the provincial roll out of HPV Primary. Details on when self-sampling will be available for population screening are not yet available.
Special cases
| Age 50 to 69 | |
| Patient Characteristics | Recommendation |
| Cervix removed with a history of biopsy confirmed high-grade squamous intraepithelial lesion (HSIL), adenocarcinoma in situ (AIS) or invasive cancer | Surveil with a provider-collected liquid-based cytology (LBC) sample from the vaginal vault at 12 months post-hysterectomy.
If HPV-negative: Routine HPV based screening at 3-year intervals (average risk) or 1-year interval (immunocompromised). If HPV-positive: Follow current HPV primary screening clinical pathway. These are interim guidelines pending updated Clinical practice guidelines. |
| History of biopsy confirmed high-grade squamous intraepithelial lesion (HSIL), adenocarcinoma in situ (AIS) or invasive cancer without total hysterectomy | Surveil with a provider-collected liquid-based cytology (LBC) sample at 12 months post discharge from colposcopy care.
If HPV-negative: Routine HPV based screening at 3-year intervals (average risk) or 1-year interval (immunocompromised). If HPV-positive: Follow current HPV primary screening clinical pathway. These are interim guidelines pending updated Clinical practice guidelines. |
| Patients on surveillance with annual Pap follow up | On their next scheduled Pap test, screen with HPV testing using a provider collected liquid-based cytology (LBC) sample.
If HPV negative: Routine HPV based screening at 3-year intervals (average risk) or 1-year interval (immunocompromised). If HPV positive: Follow current HPV primary screening clinical pathway. These are interim guidelines pending updated Clinical practice guidelines. |
| Age 65 to 69 | |
| Patient Characteristics | Recommendation |
| Has a negative HPV screen and under no active surveillance of pre-cursor abnormalities | No further screening required. |
| Inadequate screening history or has not screened in last 5 years and generally well | Screen with provider-collected LBC sample. Stop screening if result is HPV-negative. |
Download the HPV Primary Overview and FAQs for Providers
—————————————————————————————————————————————————————————————————————
HPV Self-Sampling (Cervix Self-Screening) Pilot Project
The pilot project is no longer accepting new patients or orders for self-screening test kits.
Project Background
The HPV Self-Sampling (also known as Cervix Self-Screening) Pilot Project is an Alberta Cervical Cancer Screening Program (ACCSP) initiative, funded by Alberta Health. The purpose of the project is to increase cervical cancer screening access and participation in three under-screened populations in Alberta: Indigenous, newcomer and rural/remote. In July, the eligibility criteria for the pilot project will be expanded to include patients who have not had a Pap test in the last 5 years. The project aims to develop effective and sustainable HPV self-sampling strategies, laying the foundation for potential future expansion.
Phase 2 of the project is currently underway. Community partners are engaging and recruiting eligible patients to participate in cervix self-screening.
Phase 3 is launching in May. This phase will target eligible people living in select areas in North and South Zones with low cervical cancer screening participation. Eligible people will either receive an invitation letter with information on how to order a self-screening kit OR they will receive an invitation letter with a self-screening kit.
Primary care providers may have patients who are participating in the project. You will receive lab results and a result letter for any patients who identify you as their primary care provider. All results will be available in Netcare (under microbiology). Patients can access their results through MyHealth Records or MyAHS Connect and they will receive a result letter in the mail.
Primary care providers play an important role in follow-up of abnormal results. Triage Pap testing will be required for some patients with abnormal HPV results and colposcopy referral will be required for other types of abnormal results (HPV and triage Pap). Note that this project is trialing a new colposcopy referral process aimed at expediting access to colposcopy. Please see the resources below for more information.
We encourage providers to promote the project with patients who meet the eligibility criteria Cervix Self-Screening Pilot Project – Screening For Life | Screening For Life. Patients can call 1-866-727-3926 (Monday to Friday, 8:00 am to 4:30 pm) to be assessed for eligibility and to order a self-sampling kit. Orders will be accepted until Sunday November 30, 2025.
If you have questions about the project, please email ACCSP@ahs.ca.
Clinical Pathways
HPV Self-Sampling Clinical Pathway
HPV Self-Sampling Clinical Pathway Immunocompromised Patients
Primary Care Provider Follow-Up Table
Primary Care Provider Follow-Up Table
Provider Frequently Asked Questions
FAQ for Primary Care Providers
—————————————————————————————————————————————————————————————————————
What is the HPV Test of Cure?
The HPV Test of Cure is a risk stratification tool for colposcopists. The purpose of this test is to determine which patients in colposcopy care will need ongoing follow-up in colposcopy and which can be safely discharged back to primary care for cervical cancer screening per Clinical Practice Guidelines (CPGs).
What is the role of primary healthcare providers in the HPV Test of Cure initiative?
As per the HPV Test of Cure algorithm, eligible patients in colposcopy with a negative colposcopy evaluation and HPV Test of Cure result will be discharged to primary care. Primary healthcare providers will then be responsible for their subsequent follow-up and cervical screening needs. Patients with more questions may seek primary healthcare provider assistance.
What are the benefits of HPV Test of Cure?
- To improve patient experience.
- Fewer appointments required for patients to complete the assessment and treatment cycle in colposcopy.
- An objective risk stratification tool to help determine the right care by the right provider.
How is the HPV Test of Cure different from HPV Reflex?
The test is the same; it is called different names to identify where in the care pathway the test is conducted. Currently in Alberta, the HPV test is used as a triage tool. HPV Reflex is used after an abnormal low-grade Pap test to determine referral to colposcopy while HPV Test of Cure is used after colposcopy treatment to determine if further colposcopy treatment is required.
How will primary healthcare providers be notified of patient discharge due to HPV Test of Cure status?
A communication from colposcopists will be sent to referring providers notifying them that their patient has been discharged from colposcopy services.
Who is eligible for the HPV Test of Cure?
Patients in colposcopy care who have received treatment for high-grade lesions.
Who can order the HPV Test of Cure?
Only colposcopists can order the HPV Test of Cure.
When can colposcopists start ordering the HPV Test of Cure?
Starting on September 15, 2022, colposcopists can order the HPV Test of Cure.
What happens if a primary care provider orders the HPV Test of Cure?
Any requisition for the HPV Test of Cure submitted by other providers will not be processed by the labs. Please do not order this test unless you are a colposcopist.
What happens after the HPV Test of Cure?
- Colposcopy evaluation negative + HPV Test of Cure negative = Discharge to primary care for continued cervical screening.
- Colposcopy evaluation negative + HPV Test of Cure positive = Colposcopy care + HPV Test of Cure in 6 months.
- Colposcopy evaluation positive = Treat as per colposcopy care guidelines.
Will patients receive an HPV Test of Cure result communication from the Alberta Cervical Cancer Screening Program (ACCSP)?
No, as this test is part of specialist follow-up care, communication will be from specialists to primary care providers.
Patient is asking about the HPV vaccine. What information is available?
Information on the HPV vaccine can be found here.
Need more information about the HPV Test of Cure?
Contact Alberta Health Services Screening Programs at 1-866-727-3926 or email accsp@ahs.ca.
Resources
- Evidence to Support Change of Clinical Pathway Following Colposcopy Treatment for Cervical Intraepithelial Neoplasia in Canada (jogc.com).
- HPV Test of Cure Patient Resource.
—————————————————————————————————————————————————————————————————————
HPV Reflex Testing
The HPV Reflex test is an important triage tool in the management of abnormal low-grade Pap test result. It helps determine the necessity for colposcopy in specific age groups.
- Women or people with a cervix aged 30 years and older with an ASCUS (Atypical Squamous Cells of Undetermined Significance) Pap result.
- Women or people with a cervix aged 50 years and older with an LSIL (Low-Grade Squamous Intraepithelial Lesion) Pap result.
If the HPV reflex result is positive, it usually indicates a higher risk of cervical cancer, prompting a referral to colposcopy. If HPV reflex is negative, it provides reassurance that patient can return to routine cervical cancer screening. This approach helps to reduce unnecessary colposcopy referrals. See HPV Reflex test resource for more details.